Het Anabolenboek
Willem Koert
Aede de Groot
Wageningen, 30/09/2007
1. Androstenedione
Willem Koert, Aede de Groot
The difference between food supplements and pharmacological preparations disappeared in October 1996, when the American company Osmo launched the supplement Androstene. The active principle in Androstene is the prohormone androstenedione - scientific name: 4-androstene-3,17-dione.
Androstenedione has been on the market for less then ten years. In 2004 George Bush signed a new version of the Anabolic Steroid Control Act, which equated androstenedione to anabolic steroids like methandienone and oxymetholone. Companies were not allowed anymore to sell androstenedione as a food supplement.
At first sight this gives the impression that androstenedione is a closed subject. Nevertheless we will discuss this prohormone in depth. The history of androstenedione shows characteristics that reappear continuously in many other steroids on the supplement market.
Androstenedione was not a new compound. Researchers discovered it in the thirties of the 20th century in urine, when they were looking after male hormones. With these compounds they hoped to revitalize elder man. Already in 1935 it was known that compounds were produced in the testes that stimulated sexdrive and initiated the development of male characteristics. Also at method to measure the effects of such compounds was developed. The prostate enlargement in castrated rats was measured after injection of the compounds.
In the midthirties scientists thought for a short period that androstenedione was that male sex hormone. However Ruzicka showed that this was not the case, when he found that the effect of androstenedione on the growth of the prostate was limited. Since, scientists regarded androstenedione as a "probable intermediate product in the biological formation of the male and female sexual hormones from cholesterol". [1]
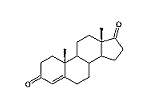 |
It was discovered that androstenedione is biosynthesized in the testes and in the adrenal gland. It is a direct precursor of testosterone, which is obtained from androstenedione after reduction of the C17 carbonyl group to a hydroxyl group. Androstenedione is also a precursor for the female sexhormone estrone (see Scheme 1). A more extensive treatment of the biosynthesis and metabolism of androstenedione is shown in Chapter 11.

Scheme 1
Not much news was published about androstenedione in the next years, but this changed in 1981 when Eastern German chemists started to study it with renewed interest. Eastern Germany or the DDR was then a communistic country that liked to distinguish itself from other countries through successes in sport. The story went that doping was used by DDR athletes on a large scale. It probably will last years before all the evidence will surface.
New doping compounds were developed by DDR chemists in secret programs as described in Stateplantheme 14.25, also called Komplex 08. Later it would become clear that in total about fifteenhundred medical docters, coaches and scientists had been involved.
At the end of the seventies the International Olympic Committee (IOC) wanted to test sportsman on the abuse of anabolic steroids and testosterone. The leader of the DDR doping project, sports doctor Manfred Hoeppner, organized a meeting of six professors, a medical doctor and a senior scientist on 24 June 1981 in Leipzig. They had to develop new doping agents that should get atround the tests of the IOC. [2] [3] [4]
Most scientists in this project worked in the Forschungsinstitut fur Korperkultur und Sport. Among them was the biochemistry professor Rüdiger Häcker, and he hit upon the idea to investigate androstenedione as doping agent. They discovered that sportsman could take it as a pill or as a spray. The latter caused a fast increase, but also a fast decrease of the concentration of androstenedione in the body, which minimized its discovery in doping tests.
Hormone producer Jenapharm, the regular supplier of doping preparations for the DDR doping program, started to produce the spray’s. They contained a solution of androstenedione and testosterone in water-ethanol and some surfactant to dissolve the steroids. The Germans discovered that the spray’s had a mental effect. Steroids like testosterone diminish stress and enhance concentration and aggression.
After the disappearance of the DDR, director Häcker decided to turn his knowledge to money. He and his colleague Claudia Mattern filed patents on the use of androstenedione, and on the use of anabolic spray’s. [5] [6] [7]
These patents finally reached the desk of the American chemist Patrick Arnold. In the US the regulations about food supplements had been widened in 1994, which gave companies more freedom. Arnold was at that time employed
 |
When Osmo introduced androstenedione in October 1996, as capsules and not as a spray, the company used details from the Eastern German patents in its advertising, to substantiate its claims on the activity of this steroid. The company itself did not investigate the activity nor the safety of androstenedione.

WO 93/21924
Androstenedione was a cash collector. It appeared in 1998 that this prohormone had increased the profits of the company by twentyfold. [8] At that time many other companies had already marketed their own androstenedione products.
This was the result from a small scandal around the popular baseball player Mark McGwire. In an interview, a reporter of Associated Press noticed that McGwire carried a bottle with androstenedione. The reporter made a note and later published a story about androstenedione that went around the world. A furious discussion started about this prohormone, whether it was legal or whether McGuire was using doping or not. Through this discussion the reputation of Andro improved enormously and the sales went up.
Patrick Arnold became famous and the magazine The Sporting News placed him among the one hundred most powerful man in sports.
When the McGwire affair hit the news, Arnold was already a much cited personality on news sites and message boards on internet where users and producers of bodybuilding supplements exchanged news and information. The chemist, an amateur bodybuilder himself, was and is held in respect for his knowledge and discussions, which he usually dominated. "There may be some chemists who know certain aspects of pharmacology more than I do", Arnold wrote some years ago, "But when you add up overall knowledge of chemistry and pharmacology, I will go up against the best of them. I know by heart how to make most every steroid."
The boards were an important vehicle for Arnold. They provided him with an intermediate to convince potential customers of the value of his products. Arnold explained for instance in the Dirty Dieting Newsletter, published by doping goeroe Dan Duchaine in 1997 and 1998, how to convert androstenedione into testosterone in your own kitchen. Just for fun, according to Arnold.
But what about the activity of androstenedione? The supplement companies have never investigated the activity nor the safety of androstenedione. To find an answer on these questions scientists started testing of androstenedione in the end of the nineties but the results were disappointing.
JAMA. 2000 Feb 9;283(6):779-82
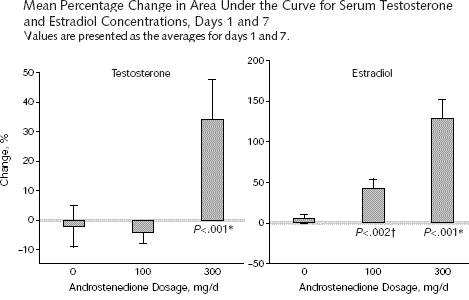
In 2000 a publication in JAMA of the Massachusetts General Hospital told that a daily dose of 100 milligram of androstenedione was not sufficient to increase the testosterone production in young man. The testosterone level was even slightly lowered. A daily dose of 300 mg of androstenedione proved to be necessary for a significant increase of the testosterone level. However, the estradiol level was raised also, and even more then the testosterone level. [9] From these results it became clear that androstenedioen also raised the chance of gynaecomastia.
A daily dose of 100 mg of androstenedione had no effect on muscle growth in young bodybuilders, as was shown by scientists of the University of Texas in 2000. [10] The possible effect of higher doses was not investigated.
An investigation from 2002 showed that daily doses of 50 and 100 mg did lead to higher testosterone levels in elder women. Interestingly, the estradiol level was not enhanced in these women. In the article only the effects in the first few hours after administration have been measured. Nothing was mentioned about longer lasting effects. [11] The conclusion of the investigatiors was that in women there is a chance on masculanising side effects.
Little is known about the long lasting use of androstenedione. An exeption may be the study of the University of Texas at Arlington from 2003, where the effects of a four week oral administration of a daily dose of 200 mg in elder man have been investigated. [12]
The results showed that the hormonal effects of the prohormone slowed down during longer periods of use. The extra testosterone, which is biosynthsised from androstenedione, disappeared quicker and quicker from the blood just as androstenedione itself. After four weeks, man who had used androstenedione, had just as much testosterone in their blood as man who did not use anything. The reason for this may be that androstenedione stimulates the production of enzymes that convert these steroids in other metabolites.
Weider Nutrition obtained in 2000 patents on prohormones with added plantsubstances like indol-3-carbinol and chrysine. [13] These additions should inhibit the transformation of testosterone and androstenedione into estradiol, thus making the prohormone more effective. However, scientists from Iowa State University later showed that the additions did not have any effect. [14]
The absence of desirable activity of androstendioen does not mean that there are no side effects. Scientists are in particular concerned about the lowering of the concentration of the beneficial cholesterol (HDL). This could enhance the chance of hart diseases. [15]
Doktors have published few cases on adverse effects of androstenedione. An exeption may be the bodybuilders that suffered from priapism for a few days, which had to be treated with medicines.
In Europe androstenedione has never been commercialized. Nevertheless, many sportshops and supplement dealers have sold the prohormone under the counter for years. Besides this 'grey supplemen' could be obtained on internet without problems. That possibility disappeared when the US government revised the Anabolic Steroids Control Act, by which androstenedione is considered as a full blown anabolic steroid. In the meantime the demand for androstenedione had already decreased. Other prohormones, which we will discuss in the next profiles, had taken its place.
[1] Biochem J. 1937 Mar;31(3):467-74.
[2] Science. 1991 Oct 4;254(5028):26-7.
[3] Clin Chem 1997 Jul;43(7):1262-79.
[4] Werner Franke. Nil Nocere – Gutachterliche Stellungnahme zur Art und zum Ausmaß der Beteiligung der Firma VEB Jenapharm und ihrer Wissenschaftler beim verbrecherischen Doping-System der DDR. Heidelberg, 22.12.2004. [PDF]
[5] Drug For Increasing The Level Of Testosterone In The Body. WO 93/21924 (9.12.1993); US Patent 5,578,588.
[6] Metering Spray Designed For Pernasal Application. WO 93/24107 (9.12.93)
[7] Androgens For Stimulating The Central Nervous System And For Treating Osteoporosis. WO 93/24128 (9.12.1993).
[8] Chicago Tribune, 13/11/2005.
[9] JAMA. 2000 Feb 9;283(6):779-82.
[10] J Clin Endocrinol Metab. 2000 Jan;85(1):55-9.
[11] J Clin Endocrinol Metab. 2002 Dec;87(12):5449-54.
[12] Br J Sports Med. 2003 Jun;37(3):212-8.
[13] US Patent 6,117,429
[14] Int J Sport Nutr Exerc Metab. 2000 Sep;10(3):340-59.
[15] J Athl Train. 2002 Jul;37(3):300-5.
[16] Ann Emerg Med. 2000 Apr;35(4):391-3.
|
|