Het Anabolenboek
Willem Koert
Aede de Groot
Wageningen, 20/09/2006
2. The Shape of Simple Carbon Compounds
Aede de Groot, Willem Koert
In the previous chapter we have described that organic chemists, biochemists and pharmacists together try to develop optimal compounds for the biological process that has to be stimulated. For this purpose first of all the active compound itself has to be found, but a very important step is also to find the place where it has to work. In the case of anabolic steroids this is the androgen receptor. A receptor is a large protein molecule with a pocket inside in which the active compound, the ligand, has to fit. When this is the case the protein-ligand complex changes its shape and becomes active. The fitting of the ligand in the pocket of its receptor is the result of an interaction on a molecular level and we will discuss the phenomena that play a role in such interactions.
For a good understanding of this interaction it will be necessary to know what a molecule is, what shape the molecule has, what a protein is, what shape the protein has and finally how the interaction between a ligand molecule and a receptor protein takes place. Before we will address these more complicated questions some basic knowledge about molecules of simple organic compounds, proteins and their interactions is necessary.
The smallest possible particle of a compound is called a molecule. Molecules are so small that they are not visible, not even with the most powerful instruments. Molecules are very small, but they do have a shape, which is known with reasonable accuracy. This is important because, as with every object, this shape determines whether the molecule will fit well, moderately, or not at all in the pocket of the receptor.
The molecules of a compound are composed of atoms. The number and kind of the atoms in a molecule are indicated by their bruto chemical formula. For testosterone this formula is C19H28O2, which means that testosterone contains 19 carbon atoms (symbol C), 28 hydrogen atoms (symbol H) and 2 oxygen atoms (symbol O). These atoms of the elements C, H and O each have their own seize and binding possibilities and together they determine the shape of the molecule of testosterone.
In the first chapter it was already mentioned that carbon (C) is the most important element in organic compounds and in all natural products. All carbon atoms have four possibilities for bonding with other atoms and in all natural products all four bonding possibilities are always used. These four bonding possibilities of C-atoms, or simply the four bonds of carbon, are pointing toward the corners of a tetrahedron and thus have mutual angles of 109.5° (see Figure 1).
The second element in testosterone is hydrogen (H). Hydrogen is the simplest element in the universe and it has only one possibility to bind with other atoms. So one C-atom with its four bonds binds with four H-atoms and in this way the molecule methane, with the bruto formula CH4, is obtained. We all know this molecule as natural gas, it is a natural product produced by methane bacteria. Because the four bonds of carbon are pointing toward the corners of a tetrahedron, the hydrogen atoms in methane are located in the same positions and in this way the shape of the molecule methane is determined as depicted in figure 1.
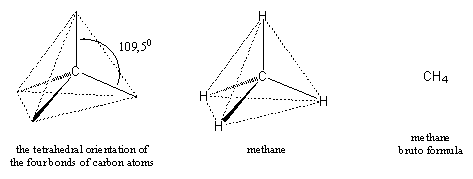
Figure 1
Carbon atoms are also very suitable to bind with each other and in this way an endless number of carbon compounds can be constructed. Carbon atoms are like lego blocks with four possibilities to connect with other units. Simple carbon compounds are the molecules ethane and ethene, both have two carbon atoms (see Figure 2). In ethane one bond of each C-atom is used to bind the other one, the other three bonds of both C-atoms are all occupied with H-atoms.
In ethene the two C-atoms are bound to each other with two bonds, a double bond, the other two places at each C-atom are occupied with H-atoms. Ethene is an important plant hormone, it itakes care of the riping of fruits and in general of aging of the plant.
It should be noticed that ethane and ethene have a very different shape. Ethane is a double tetrahedron, and has roughly the shape of a sand hour. In contrast, ethene is a flat molecule, the C- and H-atoms all lay in the same plane.
When such double bonded C-atoms are present in a larger molecule, this molecule also will be flat in the area around this double bond. There is one such a double bond present in the molecule of testosterone and three double bonds are present in the anabolic steroid trenbolone. For this reason trenbolone is a flatter molecule than testosterone.
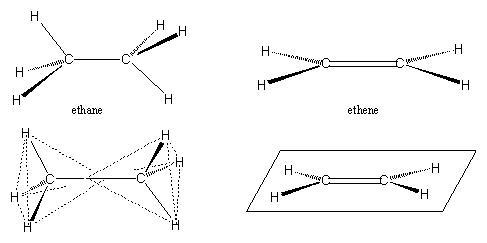
Figure 2
The third element in testosterone is oxygen (O) and the O-atom has two possibilities to bind with other atoms. Thus one O-atom binds with two H-atoms and in this way the molecule H2O is obtained, a molecule that is well known as water. The water molecule is planar and the bonds in water make an angle of 105°.
One C-atom with its four bonds binds with two O-atoms, each with a double bond. In this way the molecule CO2 is obtained, better known as carbondioxide. The two double bonds are in a direct line and therefore CO2 is a linear molecule.
Another well known molecule is ethanol or simply alcohol. C-C, C-O, C-H and O-H bonds keep the molecule together and determine its doggy shape as shown in figure 3. Ethanol can be considered as water in which one of the H-atoms is replaced by an ethyl group, a group of 2 C-atoms derived from ethane.
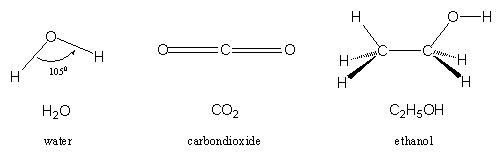
Figure 3
Four other kinds of atoms are regularly found in anabolic steroids. These are nitrogen (symbol N), and the halogen atoms fluorine (symbol F), chlorine (symbol Cl) and bromine (symbol Br).
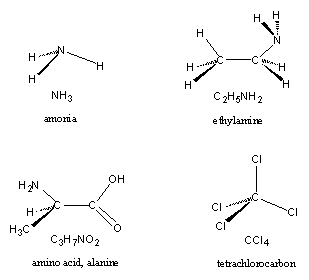 Figure 4 |
An N-atom has three possibilities to bind with other atoms and one of the simplest N-containing molecules is ammonia (NH3) (see Figure 4). When one of the H-atoms of ammonia is replaced by an ethyl group, ethylamine is obtained. N-atoms are abundantly present in proteins, which are composed of amino acids (see next Chapter). A simple example of an amino acid is alanine.
The halogen atoms F, Cl and Br all have only one bonding possibility and consequently one C-atom binds four halogen atoms, as is shown for four Cl-atoms in the molecule CCl4. A simple Cl-containing molecule is the strong acid hydrogen chloride (HCl), which is the acid in our stomach. An F-atom is found in the anabolic steroid fluoxymesterone and a Cl-atom is present in halodrol and in oral turinabol.
|
|